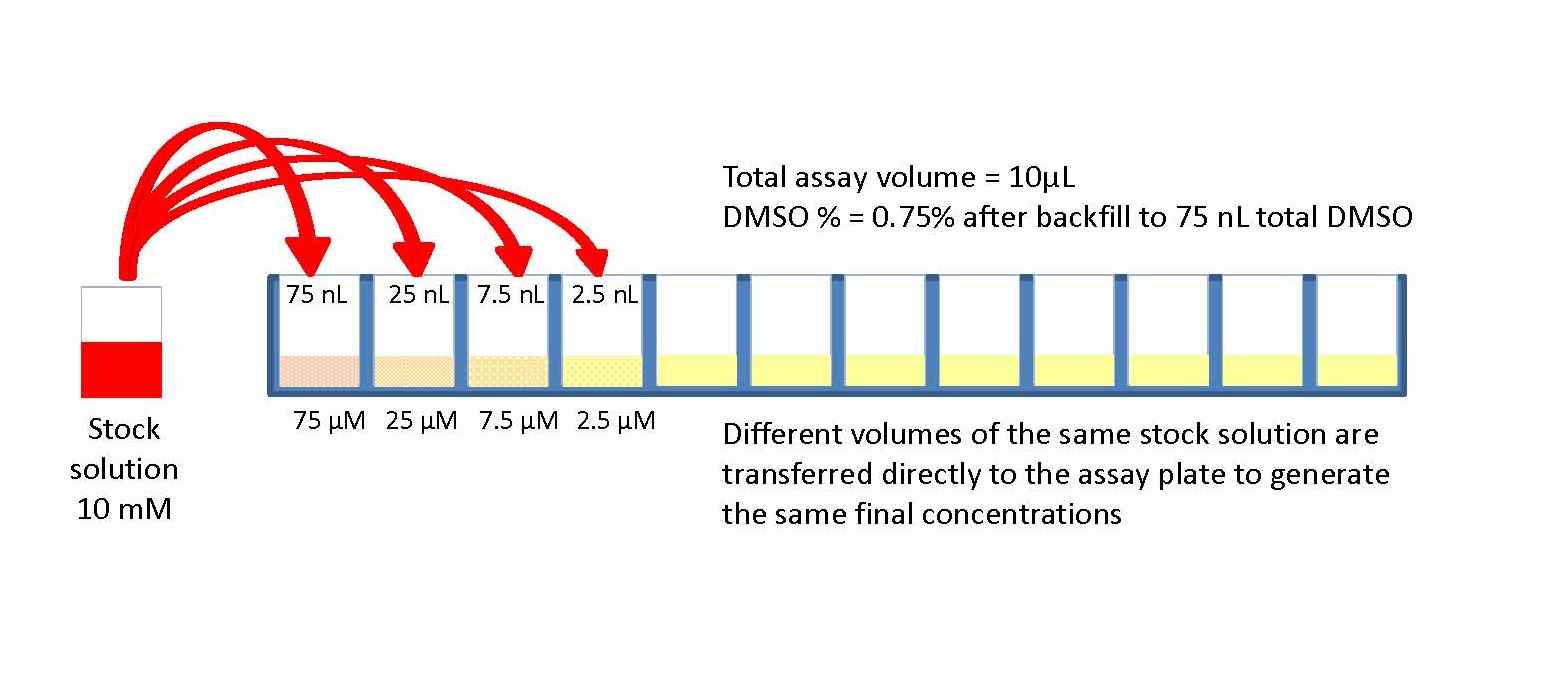
This type of dilution series is referred to as a serial dilution. 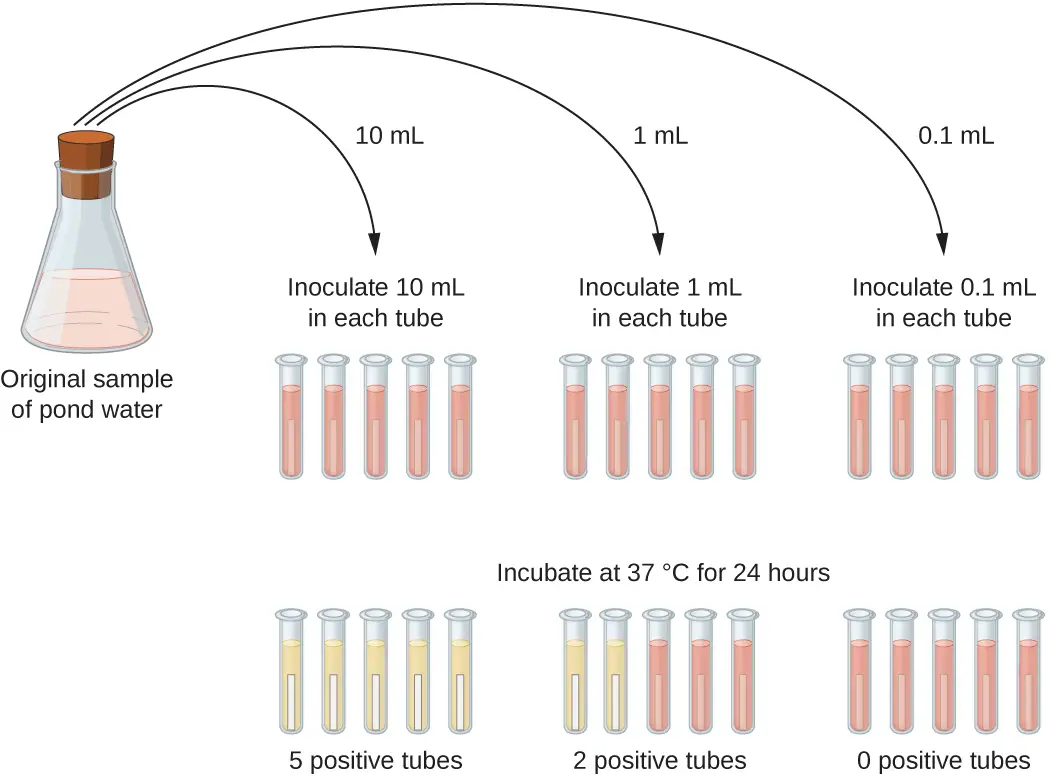
Thus: 1/100 x 1/5 1/500 Serial Dilutions Many procedures call for a dilution series in which all dilutions after the first one are the same. Clinical Chemistry: Principles, Procedures, Correlations. 1/100 dilution that was already prepared i.e., bring 1 part of the 1/100 dilution of serum in buffer up to 5 parts total volume. 'Is homeopathy a clinically valuable approach?'. 'Homeopathy: Holmes, Hogwarts, and the Prince of Wales'. Experiments in Microbiology, Plant Pathology and Biotechnology. Serial dilutions are used to accurately create highly diluted solutions as well as solutions for experiments resulting in. A ten-fold serial dilution could be 1 M, 0.1 M, 0.01 M, 0.001 M. In homeopathy, serial dilutions (called potentisation) are often taken so far that by the time the last dilution is completed, no molecules of the original substance are likely to remain. A serial dilution is the stepwise dilution of a substance in solution.Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion. Serial dilution is one of the core foundational practices of homeopathy, with 'succussion', or shaking, occurring between each dilution.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
